You are here:
- Procedures / Keratoconus / CXL
Corneal Crosslinking (CXL)
What is Keratoconus?
Keratoconus is a common cause of poor vision in young people caused by a progressive abnormality of corneal shape.
The cornea is the clear part of the front of the eye wall. You need a regular corneal curvature to help focus light accurately through the eye onto the retina.
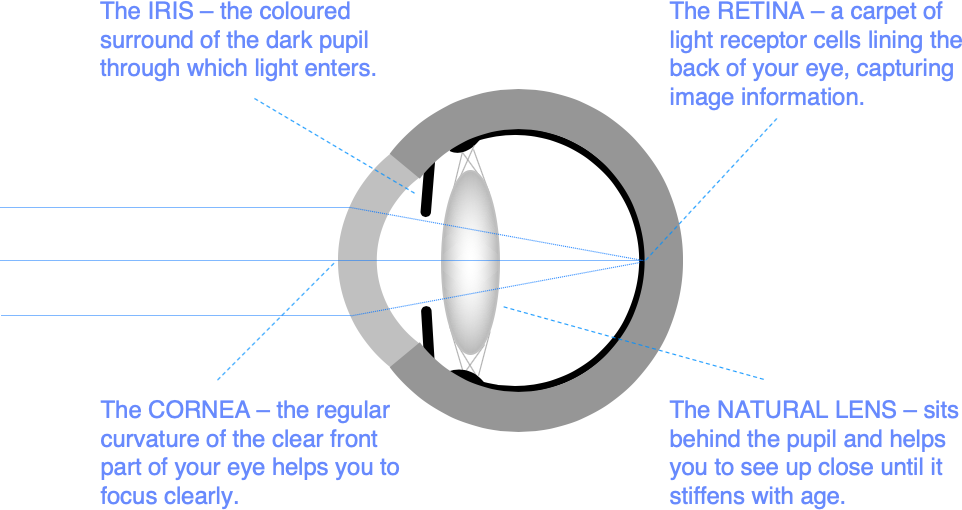
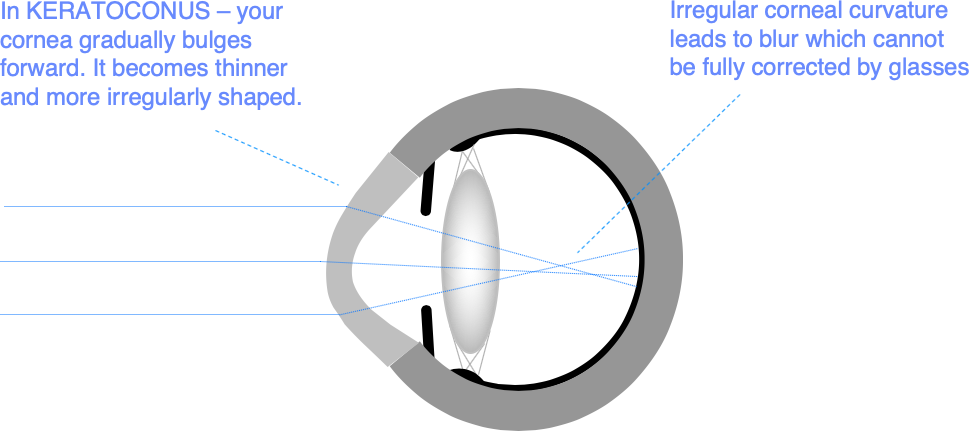
In keratoconus, your cornea gradually bulges forwards, becomes thinner, and more irregular. It typically gets worse in the teenage years and through your 20s, before stabilising in your 30s. Although it is genetically determined, only about 10% of people with keratoconus have another affected family member, and one eye is often worse affected than the other.
What is corneal crosslinking (CXL)?
Keratoconus stabilises as we get older because of a natural age-related change called crosslinking which causes the cornea to get tougher and less elastic. Crosslinking treatments are based on this natural process, and are designed to stabilise corneal shape.
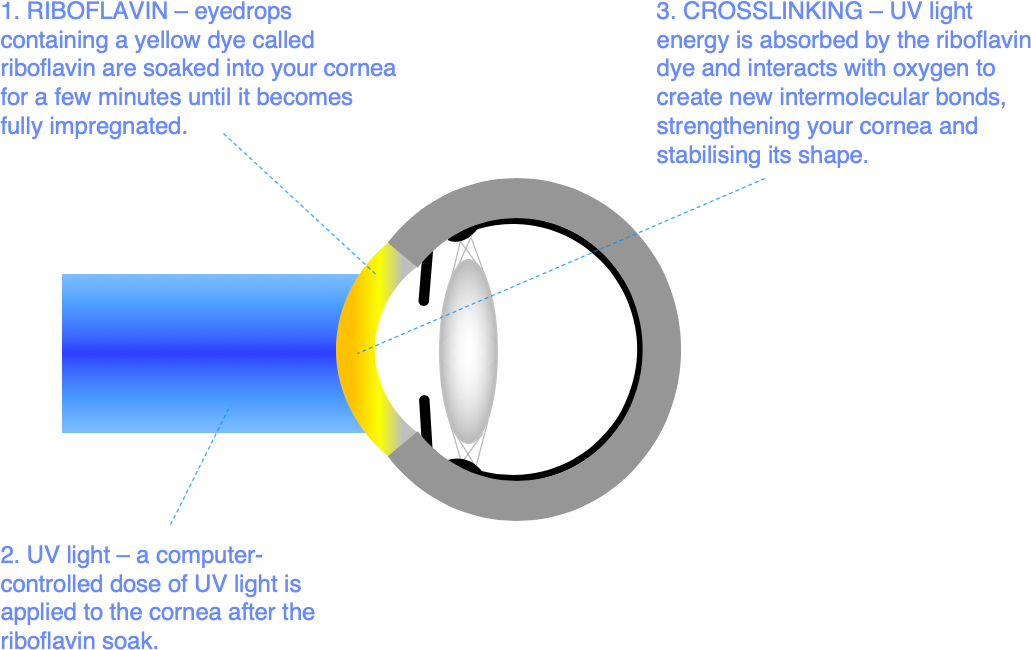
In corneal crosslinking (CXL), eyedrops containing a UV light absorbing yellow dye (riboflavin) are soaked into your cornea. Once the cornea is fully impregnated with riboflavin dye, UV light exposure in the presence of oxygen creates the conditions for new intermolecular bonds (crosslinks) to form. These additional intermolecular links strengthen the cornea, helping to stabilise corneal shape and prevent further visual loss.
CXL takes about 20 minutes per eye, and is done using eyedrop anaesthetic.
What are the benefits of CXL?
The most commonly performed types of CXL aim primarily to stabilise your corneal shape and stop your vision getting worse.
The benchmark version of the treatment is called “epithelium-off” or “Epi-off CXL.” Over 95% of people treated with Epi-off CXL are stable after treatment, and do not have any further deterioration in their vision due to keratoconus.
CXL can be repeated if necessary.
Who is suitable for CXL?
If you have keratoconus, but you are still able to see normally in glasses or soft contact lenses, CXL is often the best initial treatment option, particularly if we think your keratoconus is likely to get worse.
If you have more advanced keratoconus, but you are comfortable and seeing well in specialised contact lenses, CXL may also be your best treatment choice.
Other treatment options may be more suitable, particularly if you have more advanced keratoconus and contact lenses are not working well.
What are the alternatives?
There are good treatments for every stage in keratoconus, we will give you clear advice on your best treatment options at your keratoconus consultation.
Simple monitoring
CXL has some risks, and some patients with early stage keratoconus are already stable. So, if you are newly diagnosed with keratoconus, we often recommend a period of corneal shape monitoring using 3D scans before offering you CXL.
Risk factors for keratoconus progression are young age, non-white European ethnic background, and more advanced disease in the other eye. If you have one or more of these risk factors for progression, we may recommend CXL soon after your diagnosis rather than a period of monitoring.
From studies done before CXL treatment was available, we know that 20% of people diagnosed with keratoconus progress to the stage at which a corneal transplant is needed to restore vision within 5 years. Many more patients progress to the stage at which good vision is only possible with specialised contact lens wear. We would like to anticipate these changes, and intercept them early. So, the minimum you should do after being diagnosed with keratoconus is ensure that you have 3D corneal shape scans, at least annually, for a period of 3 years after diagnosis.
If your corneal shape has been stable for over 3 years, further change gets less likely. Corneal shape also tends to stabilise naturally as you get older. So, not everyone with early stage keratoconus needs CXL treatment.
Epi-off CXL
In current standard Epi-off CXL, the corneal skin layer (“epithelium”) is removed over a 9mm central area to help dye absorption and make the treatment more effective. It takes about 5-7 days for the skin layer to regenerate. During this time, the eye is gritty, watery, and light sensitive. The drops we give you help to ease this discomfort, but you need a week off work to recover from Epi-off CXL.
We deliver the UV light faster and at higher energies than in the original ‘Dresden protocol” form of Epi-off CXL. This shortens treatment time, and the current version of Epi-off CXL is often called “accelerated CXL”.
This version of Epi-off CXL is known to be highly effective, and is the current standard CXL treatment.
Epi-on CXL
Although the treatment itself is not painful, Epi-off CXL has a painful recovery period. Because of this, and the risks associated with corneal epithelial removal, new versions of CXL in which the corneal epithelium is not removed are being developed. These are called “Epi-on CXL”. New versions of Epi-on CXL are safe and relatively pain free in the recovery period. We do not yet know whether they are as effective as Epi-off CXL in stabilising your corneal shape. But early results are promising, and CXL can be repeated if necessary. So, if Epi-on CXL does not work for you, Epi-off CXL is a good safety net option.
Because Epi-on CXL is very low risk and relatively pain free, it is emerging as an early treatment option for people who would rather have something positive done to stabilise their corneal shape at diagnosis rather than proceeding down the standard treatment pathway of monitoring and Epi-off CXL if scans show that the corneal shape is deteriorating.
We will discuss the relative merits of alternative forms of CXL with you at your keratoconus consultation.
CuRV
Standard CXL uses a uniform UV light exposure over the cornea, and aims to stabilise corneal shape rather than improve it.
If we increase the UV light intensity over areas of your cornea that are too steeply curved, we can customise the treatment so as to flatten these areas, improving shape regularity and vision. This version of CXL is called Customised Remodelling of Vision or “CURV”. The treatment is experimental, and we do not yet know how effective the treatment is or how predictable the results are.
TransPRK/CXL
Excimer laser reshaping of the cornea can be combined safely and effectively with CXL in the right patients with the aim of improving vision and preventing further deterioration.
We use wavefront scans to create an optical map of your focus irregularity. This information is programmed into a standard laser vision correction treatment called TransPRK, and a customised laser treatment pattern is used to regularise the corneal shape. Immediately after the TransPRK stage, we can proceed with Epi-off CXL.
We performed clinical trials of TransPRK/CXL in 2015 and 2016, and we are confident that adding TransPRK to the CXL treatment is safe.
If you have problems with visual quality in spectacles or contact lenses because of your keratoconus, combined treatment can help you to get back some of what you have lost. But, in contrast to standard laser vision correction, you will normally still need glasses or contact lenses after TransPRK/CXL.
ICRS and Corneal Transplantation
If your vision in glasses is already worse than the driving standard, or your corneal shape changes are too advanced for successful contact lens fitting, it is often best to start with a treatment that gives you a fresh start with corneal shape.
Intracorneal ring segment (ICRS) implantation is a low risk procedure in which small plastic ring implants are threaded into a circular laser cut tunnel in the cornea. ICRS act to flatten the central corneal curvature. The effect on vision is variable, but about 2 patients in 3 get a significant improvement, and ICRS implantation can often save the need for corneal transplantation. ICRS implantation can be followed up with CXL or TransPRK/CXL to stabilise and fine tune the visual result.
In advanced keratoconus, corneal transplantation in which the abnormally shaped central cornea is replaced with a normal donor cornea is the best option. Corneal transplantation can be repeated if necessary, and fine-tuning revision surgical procedures can be used to optimise vision after healing. 4 out of 5 patients are able to see at the driving standard in glasses again within a year of corneal transplantation for keratoconus, and the transplant stays clear for at least 5 years in 19 out of 20 patients.
How is CXL performed?
CXL is an outpatient treatment that takes about 20 minutes. We lie you down, numb the eye surface with drops, tape your eyelashes out of the way, and use a small spring clip to help keep your eye open during treatment.
If you need treatment in both eyes, we can normally treat both eyes sequentially in the same session.
Essential steps in CXL are:
- Corneal skin layer removal – the corneal skin layer, or “epithelium”, is removed over a 9mm central zone using either a laser (in TransPRK/CXL) or by simply rubbing it away (Epi-off CXL). This step is not required for Epi-on CXL.
- Riboflavin impregnation – drops of a yellow, light-absorbing dye called riboflavin are applied, typically every 2 minutes for 10 minutes, to impregnate the cornea.
- UV light exposure – a set exposure of UV light is applied to the cornea from a specialised light source positioned in front of your eye. This stage feels a bit like going under a sun lamp, and typically takes around 8 minutes.
- Bandage contact lens placement – we finish by washing the dye away from the eye surface, applying antibiotic and anti-inflammatory eye drops, and placing a thin bandage contact lens on your eye to help comfort during healing. We will remove this contact lens for you at your postoperative review one week later.
What are the risks?
In standard Epi-off CXL, about 1 patient in 30 loses some vision because of inflammation or infection during the period in which the corneal epithelium is healing.
Many patients with keratoconus also have problems with eye rubbing and eczema type inflammation of the mucous membranes lining the eye. This is called atopic allergic blepharo-conjunctivitis. We often pre-treat the eye surface with anti-inflammatory and anti-biotic drops and tablets to optimise the eye surface prior to treatment. This helps to reduce the risk of complications during healing. We also give you intensive drop treatment in the healing period to promote comfort and to minimise the risk of infection and inflammation.
Permanent serious visual loss (vision worse than the driving standard) as a result of CXL is rare. The realistic worst scenario is that you may need a corneal transplant because of problems related to treatment.
These risks should be set against the risks of choosing not to proceed with CXL. If CXL has been recommended to you, on balance, it is your best chance of avoiding further visual loss or the need for a corneal transplant.
What are the side effects?
Side effects are problems which most patients experience to some degree after surgery. They normally improve with time, but do not always resolve completely.
Vision
Temporary blurring of vision is normal after TransPRK/CXL. Most patients find that the vision clears substantially in first few weeks after surgery. You can normally resume contact lens wear 2 weeks after treatment, but your spectacle or contact lens prescription will be altered, and you may need to wait for 3-6 months after treatment before your prescription stabilises. It is usually possible to get a temporary spectacle prescription from 2 weeks after treatment, but this may alter as your prescription stabilises with continued healing.
Eye comfort
Although the treatment itself is not painful, your eye will normally be sore for about 5-7 days after Epi-off CXL, and you will need a week off work to accommodate the recovery.
Some continued eye surface discomfort is common in the early months after most forms of eye surgery. This is usually mild after CXL, and highly variable – often affecting one eye more than the other. Treatment and prevention are based on making sure your eye surface is healthy before and after surgery. Lubricant eye drops can be helpful, and can be taken safely in addition to your other medication when required. For patients with a normal eye surface prior to surgery, lasting problems are unusual.
Eye Appearance
Red blotches are often visible on the white of the eye after any form of eye surgery. These are called subconjunctival haemorrhages, and are caused by a small leak of blood under the mucous membrane covering the white part of eye wall. Although they can be quite unsightly, red blotches are temporary, and do not affect eye health; but they can take up to 6 weeks to go away completely.
Will CXL affect my future eye care?
If you develop a new eye health problem in later life, CXL should not prevent you having successful treatment. Common eye health problems like cataract, glaucoma, diabetic retinopathy, and age-related macular degeneration can be monitored and treated as normal after keratoconus treatments including CXL, TransPRK/CXL, ICRS, and corneal transplantation.
Once your corneal shape has been stable for 3 years after monitoring or CXL treatment, further keratoconus deterioration is less likely, particularly if you are able to avoid habitual eye rubbing. But changes can occur at any age. So, we would recommend continuing with an annual eye health check under the care of your local optometrist after discharge from regular review at Moorfields. Your optometrist will be able to pick up any new problems at an early stage.
Can I have treatment to reduce the need for glasses after CXL?
Over 19 people out of 20 with keratoconus who are treated with CXL have a stable corneal shape from 6 months after treatment. From that stage, if you are heavily dependent on contact lenses or glasses, and particularly if you have a high spectacle prescription or if your spectacle prescription is much stronger in one eye than the other, you may wish to look at alternatives such as ICL implantation.
ICL implantation involves the implantation of a soft, corrective lens just behind the iris. It is commonly used to correct high levels of short sight and astigmatism. ICLs are a safe option for keratoconus patients, but they can only correct the need for glasses. If your vision in glasses is poor, you may need TransPRK to regularise the optical shape of your cornea as a preliminary step.
We can discuss these follow-up treatment options with you at any stage after your 6-month postoperative consultation.
How can I reduce the risk of problems?
You can eat and drink normally before treatment, and should take any regular medication as usual.
Stay as relaxed as you can during the procedure, and try to keep your head still after we have positioned it comfortably for you. Look up to the target light above your eye during the UV light application, this helps to keep your eye in the best position for treatment.
Most people are anxious prior to treatment. We are used to this, and we will coach you through the procedure, encouraging you at every stage. Keep your breathing calm and tell us if you need a break. An anti-anxiety, muscle-relaxing drug such as diazepam can be helpful, particularly if you tend to squeeze your eyes shut when they are being touched. Let us know if you are worried, and we will help you to relax.
A spring clip is used to hold the eyelids apart during treatment, and the UV light source has an eye-tracker built in. So, you should not worry too much about moving or blinking during the procedure. But try to listen to instructions and keep your eyes on the flashing fixation light when asked to do so.
Rest with your eyes closed when you can after treatment, and use the lubricant drops you will be given frequently. It is also nice to listen to the radio, podcasts or talking books to take your mind off the discomfort you will feel in the first few days.
You can wash and shower normally once the bandage contact lenses have been removed one week after surgery. In the meantime, try to keep the skin around the eyes clean, and remove dried mucous from the eyelashes with moist cotton wool pads. Sports can be resumed as soon as you feel comfortable again, but you should not swim until your bandage contact lenses have been removed.
Set a smart phone reminder and use the antibiotic and anti-inflammatory drops as prescribed to help the eyes to heal well. It is good to leave at least 2 minutes between different types of eye drop so that they each absorb well before the next drop is applied. If you miss the first time or you are not sure, applying a second eye drop is no problem.
Some variability of vision and comfort is normal in the early weeks after surgery, and patience is required. But you should not be afraid to contact us if you have any concerns, or if you have an injury to the eye. You should contact us without delay if you have increasing pain, light sensitivity, redness, blur or an injury to the eye followed by pain, blur or watering. You will be given Mr Allan’s mobile phone number for emergency contact after surgery. The practice management and the laser nursing team are also available for informal advice. The Eye Casualty Department at Moorfields Eye Hospital is open for emergency review 24 hours a day, 7 days a week.
You may not be aware of a problem that requires treatment in the healing phase. So, make sure you attend your review appointments even if your eyes feel good.
Appointments and enquiries
To arrange a private consultation, please telephone Angelique Thomas on 020 7566 2156 or 07484 081815 (or from outside the UK +44 20 7566 2156 or +44 7484 081815) or email moorfields.ballan@nhs.net
For NHS treatment with Mr Allan’s team at Moorfields, you will need a referral from your GP or Ophthalmic Surgeon. Referrals should be addressed to Mr Bruce Allan, Consultant Ophthalmic Surgeon, Moorfields Eye Hospital, City Rd, London EC1V 2PD. If you have any difficulty with your NHS referral, please call Barbara Stacey, NHS secretary to Mr Allan on 020 7566 2320, or email barbarastacey@nhs.net.